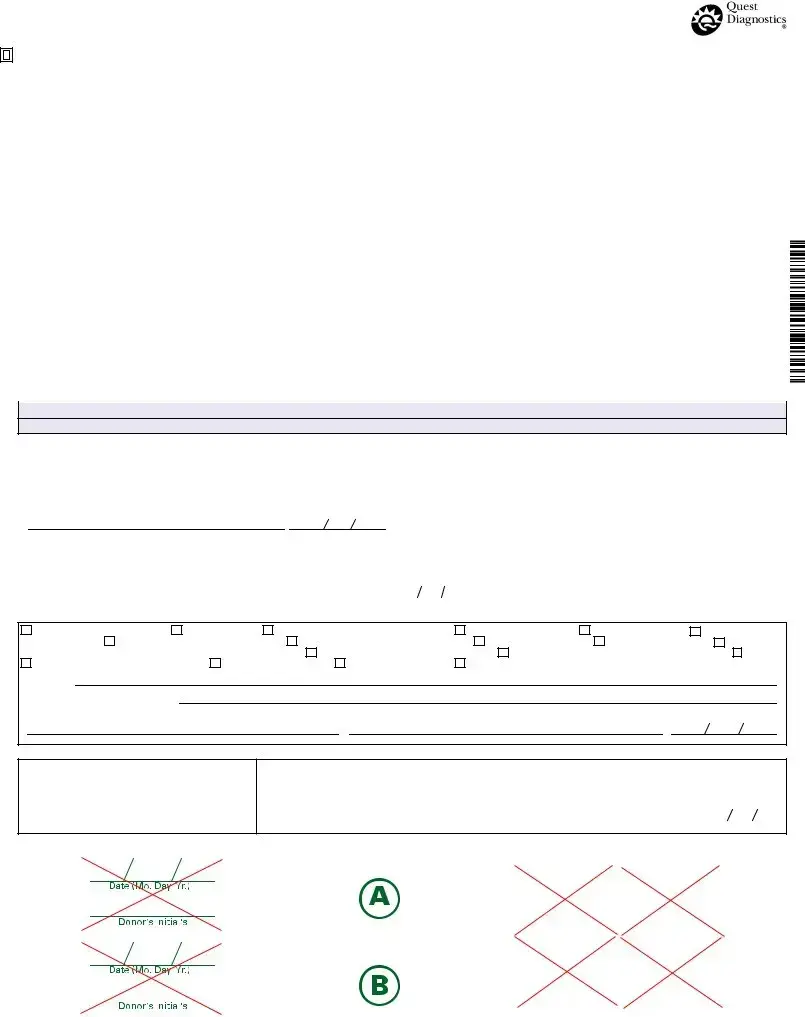
What is the purpose of the Drug Screen form?
The Drug Screen form is used to document the process of collecting and testing a urine sample for drugs. It ensures compliance with federal regulations and provides a clear record of the chain of custody for the specimen. This form is essential for employers, especially those in safety-sensitive industries, to maintain a drug-free workplace and to comply with testing requirements set by various authorities.
Who completes the Drug Screen form?
The form is primarily completed by the collector or an employer representative. This individual is responsible for gathering the specimen, ensuring proper labeling, and maintaining the chain of custody. Additionally, the Medical Review Officer (MRO) may also have a role in completing parts of the form, particularly regarding the interpretation of test results.
What information is required on the Drug Screen form?
Key information includes the employer's name and address, the MRO's contact details, the donor's Social Security Number or employee ID, the testing authority, the reason for the test, and the specific drugs to be tested. The form also requires details about the collection site and the collector's information, ensuring that all necessary parties are clearly identified.
What steps are involved in the collection process?
The collection process involves several steps. First, the collector verifies the donor's identity and reads the specimen temperature within four minutes to ensure it falls within the acceptable range. Next, the specimen is collected, labeled, and sealed. The donor then initials the seals to confirm their agreement with the process. Finally, the collector documents the chain of custody, ensuring the specimen is securely handled until it reaches the testing facility.
What happens if a specimen is rejected for testing?
If a specimen is rejected, the form will indicate the reason, such as being adulterated, substituted, or invalid. The testing facility must document these reasons clearly. This information is crucial as it informs the employer and the donor about the status of the test and any necessary follow-up actions that may be required.
How is confidentiality maintained during the testing process?
Confidentiality is a priority throughout the drug testing process. The Drug Screen form is designed to limit access to sensitive information. Only authorized personnel, such as the collector, MRO, and designated testing facility staff, have access to the results. Additionally, results are typically reported in a manner that protects the identity of the donor, ensuring compliance with privacy regulations.
What should I do if I have questions about the Drug Screen results?
If you have questions regarding the Drug Screen results, it is important to contact the MRO listed on the form. The MRO is responsible for interpreting the results and discussing any potential implications with the donor. They can provide clarity on the findings and guide individuals on the next steps, especially if a positive result is reported.