What is the purpose of the Controlled Drug Log form?
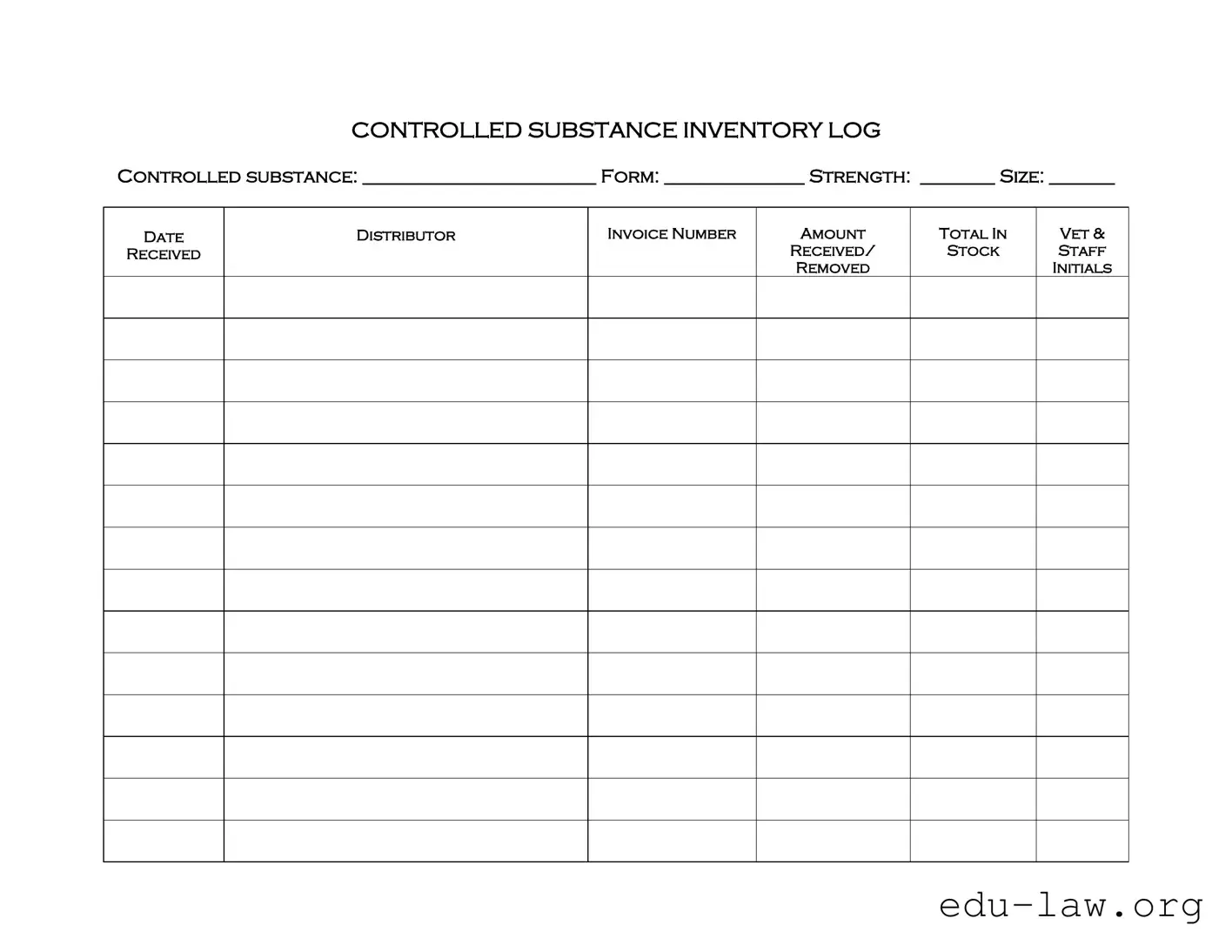
The Controlled Drug Log form is designed to track the inventory of controlled substances within a facility. It helps ensure accurate record-keeping of the drugs received, stored, and dispensed. By maintaining this log, staff can monitor usage and comply with regulatory requirements, thus promoting safety and accountability in handling controlled substances.
What information is required on the Controlled Drug Log form?
The form requires several key pieces of information. You must fill in the name of the controlled substance, its form (e.g., tablet, liquid), strength, and size. Additionally, each entry must include the date of receipt, the distributor's name, the invoice number, the amount received, and the total amount in stock. Staff initials are also needed to confirm the removal of any substances from inventory.
Who is responsible for maintaining the Controlled Drug Log?
The responsibility for maintaining the Controlled Drug Log typically falls to designated staff members, such as pharmacists or veterinary technicians. These individuals are tasked with ensuring that the log is updated accurately and regularly. However, all staff handling controlled substances should be aware of the log's contents and procedures to promote accountability and compliance.
How often should the Controlled Drug Log be updated?
The Controlled Drug Log should be updated each time a controlled substance is received or removed from inventory. This means that any time a new supply arrives or a dose is dispensed, the log must reflect these changes immediately. Regular audits of the log are also recommended to verify accuracy and ensure compliance with regulations.
What should be done if an error is found in the Controlled Drug Log?
If an error is identified in the Controlled Drug Log, it is important to correct it promptly. The staff member responsible should strike through the incorrect entry with a single line, write the correct information next to it, and initial the correction. This method maintains the integrity of the log while providing a clear record of changes. If the error is significant, it may be advisable to notify a supervisor for further guidance.